Initialising ...
Initialising ...
Initialising ...
Initialising ...
Initialising ...
Initialising ...
Initialising ...
 - and K-montmorillonite
- and K-montmorilloniteKawakita, Ryohei; Saito, Akito*; Sakuma, Hiroshi*; Anraku, Sohtaro; Kikuchi, Ryosuke*; Otake, Tsubasa*; Sato, Tsutomu*
Applied Clay Science, 231, p.106722_1 - 106722_7, 2023/01
Times Cited Count:1 Percentile:21.06(Chemistry, Physical)Tsugawa, Kiyoto*; Hayakawa, Sho*; Okita, Taira*; Aichi, Masaatsu*; Itakura, Mitsuhiro; Suzuki, Katsuyuki*
Computational Materials Science, 215, p.111806_1 - 111806_8, 2022/12
Times Cited Count:2 Percentile:29.01(Materials Science, Multidisciplinary)Yotsuji, Kenji*; Tachi, Yukio; Sakuma, Hiroshi*; Kawamura, Katsuyuki*
Genshiryoku Bakkuendo Kenkyu (CD-ROM), 29(2), p.63 - 81, 2022/12
The understanding of the swelling phenomenon of montmorillonite is essential to predict the physical and chemical behavior of clay-based barriers in radioactive waste disposal systems. This study investigated the key factors controlling crystalline swelling behavior of montmorillonite with different interlayer counter-ions by molecular dynamics (MD) simulations. On the basis of the comparisons between MD simulated and experimental results, the water content in the interlayer in five homoionic (Na , K
, K , Cs
, Cs , Ca
, Ca and Sr
and Sr ) montmorillonite was strongly correlated to the hydration number and the preference of an outer- or inner-sphere complex of each counter-ion. The detailed analysis for these results offer insights that the hydration number is controlled by the hydration free energy, the volume and the distribution of each interlayer counter-ion. The systematic MD simulations with virtually variable parameters clarified that the hydration free energy and the charge of interlayer counter- ions compete as influencing factors, and the control the formation rate of an outer-sphere complex of each counter-ion. The empirical relationships between these key factors will allow essential insights into predicting the swelling behavior of montmorillonite with different interlayer counter-ions.
) montmorillonite was strongly correlated to the hydration number and the preference of an outer- or inner-sphere complex of each counter-ion. The detailed analysis for these results offer insights that the hydration number is controlled by the hydration free energy, the volume and the distribution of each interlayer counter-ion. The systematic MD simulations with virtually variable parameters clarified that the hydration free energy and the charge of interlayer counter- ions compete as influencing factors, and the control the formation rate of an outer-sphere complex of each counter-ion. The empirical relationships between these key factors will allow essential insights into predicting the swelling behavior of montmorillonite with different interlayer counter-ions.
Okumura, Masahiko; Kerisit, S.*; Bourg, I. C.*; Lammers, L. N.*; Ikeda, Takashi*; Sassi, M.*; Rosso, K. M.*; Machida, Masahiko
Journal of Environmental Radioactivity, 189, p.135 - 145, 2018/09
Times Cited Count:51 Percentile:87.2(Environmental Sciences)no abstracts in English
 -butyl phosphate in organic and aqueous environments and its relevance to nuclear extraction processes
-butyl phosphate in organic and aqueous environments and its relevance to nuclear extraction processesMu, J.*; Motokawa, Ryuhei; Williams, C. D.*; Akutsu, Kazuhiro*; Nishitsuji, Shotaro*; Masters, A. J.*
Journal of Physical Chemistry B, 120(23), p.5183 - 5193, 2016/06
Times Cited Count:27 Percentile:60.26(Chemistry, Physical)Suzuki, Satoru; Kawamura, Katsuyuki*
JNC TN8400 2001-005, 41 Pages, 2001/04
A correlation between molecular structure and a vibrational spectrum of interlayer water in Na-smectite was investigated by means of Molecular Dymamics (MDs) simulations. Detailed comparison of simulation results with IR spectroscopic observations for the water-smectite system indicated good agreement. Internal vibrational spectra of water were obtained by the Fourier transformation of velocty auto-correlation function of hydrogen atom. A stretching vibrational spectrum of interlayer water consisted of a broad band with a peak top around 3400cm and a sharp peak around 3650 to 3700cm
and a sharp peak around 3650 to 3700cm . The fomer broad band was assigned to O-H vibrations between water molecules as bulk water, while the latter band was attributed to O-H ones oriented to siloxane surface through hydrogen bonding. The hydrogen bond distance, determined as the shortest O-O distance by the radial distribution function (RDF), revealed that hydrogen bond distance between water and siloxane surface (O
. The fomer broad band was assigned to O-H vibrations between water molecules as bulk water, while the latter band was attributed to O-H ones oriented to siloxane surface through hydrogen bonding. The hydrogen bond distance, determined as the shortest O-O distance by the radial distribution function (RDF), revealed that hydrogen bond distance between water and siloxane surface (O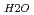 -O
-O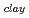
 3.0
3.0  -O
-O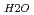 = ca. 2.8 AA ). These results suggested that interaction between water molecule and siloxane surface weaker than that between water molecules, although they were forced to be oriented.
= ca. 2.8 AA ). These results suggested that interaction between water molecule and siloxane surface weaker than that between water molecules, although they were forced to be oriented.
Pinak, M.
Proceedings of 10th International Congress of the International Radiation Protection Association (IRPA-10) (CD-ROM), 5 Pages, 2000/00
no abstracts in English
Okamoto, Yoshihiro; Kobayashi, Fumiaki; Ogawa, Toru
Journal of Alloys and Compounds, 271-273, p.355 - 358, 1998/00
Times Cited Count:40 Percentile:86.02(Chemistry, Physical)no abstracts in English
 with molecular dynamics method
with molecular dynamics methodSato, Isamu; Matsumoto, Taku; Koyama, Shinichi; Arima, Tatsumi*
no journal, ,
Diffusion coefficients of fission product elements, Zr, Nd, Ba and Sr in UO crystal which are substituent cations for U were evaluated with molecular dynamics. The diffusion coefficients of Nd and Ba/Sr which are trivalent and divalent cations are greater than those of U and Zr that are tetravalent cations.
crystal which are substituent cations for U were evaluated with molecular dynamics. The diffusion coefficients of Nd and Ba/Sr which are trivalent and divalent cations are greater than those of U and Zr that are tetravalent cations.
Matsui, Naoki*; Okubo, Takahiro*; Tachi, Yukio; Sugiura, Yuki; Yotsuji, Kenji*
no journal, ,
no abstracts in English
Matsui, Naoki*; Okubo, Takahiro*; Tachi, Yukio; Yotsuji, Kenji*; Sugiura, Yuki
no journal, ,
no abstracts in English